Photoresists & Specialty Chemicals
SU-8 Surface Modification – Functionalization
Part of the Applications Notes Line
Microstructures fabricated from SU-8 photoresist have excellent mechanical properties, thermal stability, etch resistance and are chemically stable. SU-8 is also highly transparent under near UV and visible light. These properties make it ideal for microfluidic and bioMEMS applications. Yet native SU-8 is also highly hydrophobic and has a low surface energy. Since water use is also required in these devices, it would imply that microfluidic systems fabricated from native SU-8 require active pumping. Surface modification eliminates that need.
Surface Modification: The polymer surface has to be rendered hydrophilic to enhance capillary flow. To this end, wet chemical or plasma-based processes may be used. Oxygen plasma activation of polymer surfaces is widely used to achieve a highly hydrophilic SU-8 surface as a result of CO and COO groups formed on the surface. This yields completely wettable surfaces with water contact angles of less than five degrees. A plasma treatment increases surface energy and often increases the surface roughness.
Functionalization: Methods for SU-8 surface functionalization, especially for bioanalytical applications, are described in the recent literature. For example, functional films such as enzyme-immobilized layers or biocompatible layers have been attached to SU-8. Biomolecules on SU-8 are of interest because of their application in bioMEMS, biomolecule immobilization related assays, biosensors, membrane bioreactors, clinical diagnostics, molecular biology, agriculture, environmental science and the chemical / biochemical industry.
SU-8 Series: Benefits/Attributes
- High aspect ratio imaging
- 0.5 to >200µm in a single coat
- Superb chemical and temperature resistance
- Optical transparency
- Photolithography is more cost effective compared to Si and glass micromachining.
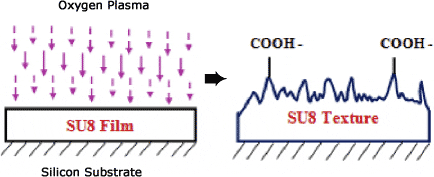
Exposure of SU-8 thin/thick film to O2 plasma.
Diagram: R.A.Singh ME Dept., Nat. U of Singapore
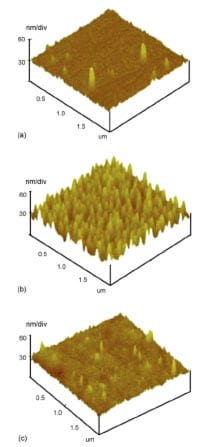
AFM pictures of SU-8 surface
(a) before surface modification,
(b) after surface modification and
(c) after antibody immobilization.
Source: M. Joshi et al. / Biosensors & Bioelectronics 22 (2007) 2429-S2435






